In 2013, my team from MerLion Pharmaceuticals in Singapore, in collaboration with Martin Lear (then at NUS), published the structure of a new acyl tetrapeptide, which was an inhibitor of an antimalarial cysteine protease drug target named falcipain-2, in The Journal of Antibiotics. We named this compound falcitidin. In our work, a small amount of a relatively pure active fraction was secured using bioassay-guided isolation after several fermentation attempts and media changes. The planar structure was elucidated by NMR and MS/MS analysis but attempts to isolate further material for biological testing were hampered by an inconsistent production and a low yield (< 0.1 mg/L). As a consequence, we decided to use an alternative approach to fermentation. First, the absolute configuration was determined by Marfey’s analysis and then the structure was confirmed using total synthesis to be isovaleric acid-D-His-L-Ile-L-Val-L-Pro-NH2. We also explored some preliminary SAR that was published in Tetrahedron Letters the next year.
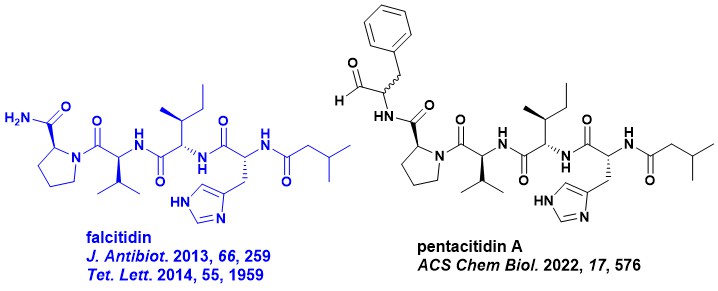
This is where the story was left until a paper in ACS Chemical Biology was recently published. This team led by Armin Bauer and Till Schäberle used molecular networking to identify over 30 naturally occurring falcitidin analogues from 25 different strains of the bacteria Chitinophaga sp. The team also investigated their biosynthesis. An example of a more potent analogue was pentacitidin A, which is shown above. Interestingly, synthetic falcitidin was not active when tested at 50 uM in their assays and they hypothesise that this could be due to different assay systems. Please read their in-depth study for further details.
My take home points:
- Bioassay-guided isolation can be difficult on occasions but is important as it can unveil new pharmacophores. Don’t give up easily if the biological signal is clear, but always use dereplication to identify known actives and to group ‘like extracts’.
- Molecular networking is a very powerful tool for identifying analogues as clearly demonstrated in this ACS Chem. Biol. paper.
- Analogue isolation and synthesis are important that led to a deeper understanding of the biological activity and potential utility of these compounds.