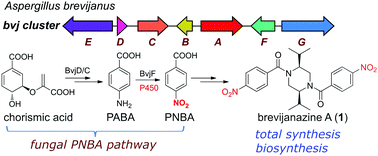
I wanted to tell you about some work I was involved with that was recently published in Chemical Communications. I was able to solve the structures of two new para-nitrobenzoic acid containing piperazines, which were named brevijanazine A and B, isolated from the fungus Aspergillus brevijanus (NRRL 1935).
The NMR data of the natural products were broad and had multiple sets of resonances due to slow conformational changes of the amide bond(s). The NMR data of the p-nitrobenzoic acid moieties, which are relatively rare in nature, were consistent with those reported for other natural products such as waspergillamide A. This is where the rest of the team (from Microbial Screening Technologies, Macquarie University, The University of Western Australia and Sun Yat-sen University) went into action and the structure was confirmed though X-ray analysis and total synthesis. Probably one of the most interesting aspects was the heterologous biosynthesis, precursor feeding and in vitro microsomal assays that showed that a cytochrome P450 oxygenase converts p-aminobenzoic acid to p-nitrobenzoic acid. It wasn’t too long ago that manipulating the genetics of fungi seemed like a distant dream.