I wanted to share a recent publication on the discovery of gargantulide B and C, which are complex glycosylated 52 membered macrolactones with antibacterial activity. The gargantulide story started when I was leading the Natural Product Chemistry at MerLion Pharmaceuticals in Singapore around 2006. We were very interested in discovery new antibacterials and identified an extract from a Streptomyces sp. that had Gram-positive activity against MRSA. In this strain, gargantulides could be produced in high titres and the major component had a molecular formula of C105H200N2O38. With a molecular formula like this, the structure was always going to be challenging. It ended up being the first structure that I couldn’t assign a planar structure.
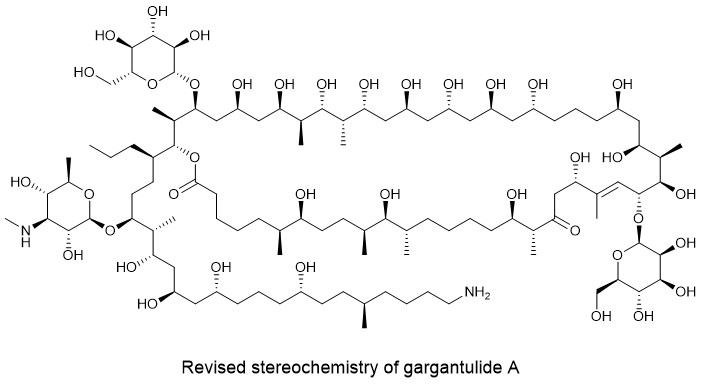
We then approached Bill Gerwick (Scripps Institute of Oceanography/UCSD) and ask him if his team was up for solving a very complex structure! It was indeed challenging with a lot of overlapping NMR resonances – even at 900 MHz. The structure was finalised a few years later by Jung-Rae Rho, who was visiting Bill’s labs from Kunsan National University. His structure is a tour de force of structure elucidation and we published the structure of gargantulide A in Organic Letters in 2015. The paper even made it into Chemical and Engineering News (p 6, 16 March 2015) with the apt title “Newfound antibiotic is gargantuan and unfortunately deadly”. Initially, we were excited at MerLion to have identified a new antibacterial class with activity against both MRSA and Clostridioides difficile, but garganulide A was found to be toxic during preliminary PK studies. Unfortunately, this is how drug discovery often turns out.
Back onto the recent Organic Chemistry Frontiers report by a team from the Fundación MEDINA in Spain and The Novo Nordisk Foundation Center for Biosustainability in Denmark. They identified an Amycolatopsis sp. that produced gargantulides. They were able to unravel and then use the biosynthetic gene cluster (very large!), along with NMR, to help confirm the structures and stereochemistry of two analogues gargantulide B and C. This team were then were able to confirm our planar structure of gargantulide A and revise some stereochemical centres. The new analogues also had antibacterial activity. Awesome work!
When asked about my “best papers” in a job interview once, the gargantulide A paper definitely made the few! It was great to see further work in this area.
Gargantulide A paper link and Gargantulide B and C paper link.