For something different…
I recently listened to Marc Andreessen’s March 2026 conversation on the David Senra‘s podcast (Spotify and YouTube), and one section stopped me in my tracks: his deep dive into how Elon Musk manages. Marc doesn’t just talk about Elon as an inventor or showman. He frames Musk as “maybe the greatest manager of our era” – a modern Henry Ford whose companies are extensions of his mind, will, and operating system. In Marc’s telling, Tesla and SpaceX are not just corporations; they are founder-run machines designed around a very specific management code.
I’ve started thinking about that code as “The Elon Code” – and how much of it could (and perhaps should) be ported into biotech.
Founder Logic vs Manager Logic
One of Marc’s central points is the contrast between founder logic and manager logic.
Manager logic is optimized for stability: layers of reporting, committees, dashboards, and process. Founder logic is optimized for forward motion: direct engagement with the product, technology, and people; continuous redesign of the system; and very little patience for ritualised “management theatre.” Marc’s claim is that Elon represents the strongest living example of founder logic at industrial scale. He compares him to Henry Ford: someone who doesn’t just run a company, but rewrites the rulebook of an industry.
In this view, the company and the founder are effectively one system. You don’t “swap in” an interchangeable manager to run SpaceX any more than you could have swapped in a random executive to run Ford in 1914.
Pillar 1: Hands-On, Engineer-First Leadership
The first element of the Elon Code is that the CEO is a technical peer, not a presentation layer. Elon doesn’t only read slides about rockets and cars; he participates directly in design discussions, engineering reviews, and system-level decisions. Engineers are not three (or 12) layers removed from the person in charge – they’re in the room with him, arguing about designs and constraints.
That has two big effects: (1) It sets an extremely high bar for technical depth in leadership, and (2) It creates positive selection: strong engineers want to work where the person at the top is genuinely engaged with their craft, not just their status reports.
In practice, this flips the usual hierarchy. Instead of engineers being downstream from management, management is built around enabling and challenging the best engineers.
Pillar 2: Technical Truth Over Narratives
Marc also emphasizes Elon’s obsession with truth at the source.
Large organizations tend to accumulate what he calls “compounding lies”: every layer summarises, sanitises, and spins reality just a little bit. By the time the CEO sees a dashboard, the mess has been smoothed into a narrative. Elon’s countermeasure is simple but radical: (1) Skip as many layers as possible, (2) Talk directly to the people doing the work, and (3) Look at the real system – the line, the rocket, the code, the data – not just the slide deck.
In other words: physics, code, and factory facts beat PowerPoint narratives. The cultural message is clear: don’t tell me a story, show me the system.
Pillar 3: Systematic Bottleneck Removal
Another key behaviour Marc highlights is Elon’s ritual of systematic bottleneck removal.
The idea is straightforward: Continuously identify the single biggest constraint on progress or throughput. Go there physically or mentally – the factory line, the subsystem, the team. Work on it personally until the constraint moves. Repeat.
If you do that every week, the CEO effectively becomes a velocity multiplier: over a year, you’ve helped solve ~50 of the biggest problems in the company, not by delegating them into a bureaucracy, but by reshaping the system around them. This is very different from the traditional CEO calendar filled with reviews, ceremonies, and low-stakes meetings. The work of leadership is reframed as: “Find the constraint, fix the constraint.”
Pillar 4: “Milli-Elon” Intensity
Marc also introduces a memorable concept: “milli-Elon” as a tongue-in-cheek unit of intensity.
Elon is set at 1,000 milli-Elons, while most people operate at single-digit milli-Elons. The actual number doesn’t matter. What matters is the idea: there is an extreme, sustained level of drive and pain tolerance behind the outcomes we see. Importantly, this isn’t just about long hours. It’s about: Willingness to confront uncomfortable truths.
Staying with hard problems long past the point most people give up. Keeping the pressure on systems, processes, and people until they change. Even operating at 50 or 100 milli-Elons – a small fraction of that bar – would still constitute intense leadership in most organizations.
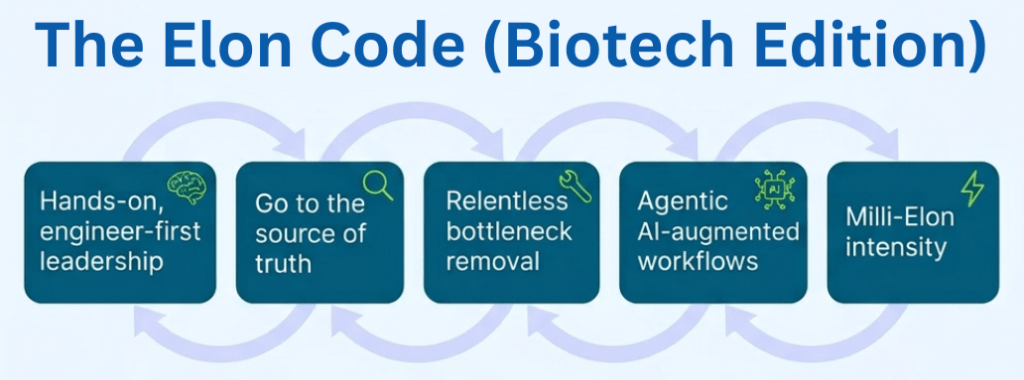
Pulling It Together: The Elon Code as a Loop
If you put these elements together, you get a simple loop:
- Hands-on, engineer-first leadership – the founder is in the technical trenches.
- Go to the source of truth – bypass reporting theatre, look at the real system.
- Relentless bottleneck removal – always work on the hardest constraint.
- Repeat at milli-Elon intensity – keep the loop running far longer and harder than average leaders.
That loop is Marc’s answer to the question: Why do companies like Tesla and SpaceX do things incumbents literally can’t? It isn’t just the ideas or the capital. It’s the operating system.
Can the Elon Code Work in Biotech?
This is where it gets interesting for our world. Biotech is full of:
- Complex, multi-step workflows (wet lab → data → analysis → reporting).
- Deep technical constraints (biology, chemistry, manufacturing).
- Heavy regulatory overhead and legacy processes.
On paper, that looks like a “managerial” environment. In practice, I think it’s primed for exactly the kind of founder logic Marc is describing. Here’s how the Elon Code might translate:
- Hands-on leadership in the stack
CSOs, CTOs, and technically fluent founders who can move between cell culture, sequencing, ML models, and regulatory conversations – not just sit on top of them. - Truth at the bench and in the data
Senior leaders who regularly go to the lab, read raw data, and talk directly to the people running experiments and pipelines – not only to program managers. - Bottleneck hunting as a habit
Treating “what is today’s biggest constraint?” as a standing agenda item: it might be an assay, a pipeline, a QC step, a regulatory interaction, or a data-access issue. - Intensity where it matters
Not performative overwork, but real willingness to push through ambiguity, slow cycles, and organisational friction to get a therapy, platform, or dataset over the line.
Layering Agentic AI on Top
Where I think biotech can add a twist to the Elon Code is in the use of agentic AI – systems that don’t just answer questions but can act, orchestrate tools, and help run workflows. Imagine applying the same loop with AI as an amplifier:
- Instrument workflows end-to-end
Agentic AI agents sit across wet-lab automation, LIMS, bioinformatics pipelines, and documentation systems, continuously collecting signals about delays, errors, and rework. - Surface real bottlenecks, not just anecdotes
Instead of relying on whoever complains loudest, AI flags where cycle times are spiking, where quality drops, or where handoffs repeatedly break. - Propose and test fixes
Agents can suggest protocol changes, re-order work queues, adjust resource allocation, or even auto-draft SOP updates and documentation for human review. - Close the loop faster
Leaders still make the judgment calls, but the “find the bottleneck → experiment with fixes → remeasure” cycle becomes much faster and more data-driven.
In other words: relentless bottleneck removal stays human-led, but becomes AI-accelerated.
A Practical Question for Biotech Leaders
You don’t need to be Elon – or want to be – to borrow some of this playbook. A few important questions worth asking inside any biotech company:
- How often do our senior leaders actually see the real system (bench, pipeline, manufacturing, safety data) rather than just slide decks?
- Do we have a shared, explicit answer to: “What is the biggest bottleneck in our work right now?”
- Are we designing roles and org structure so that the best scientific and technical talent can work with leadership on hard problems, rather than just reporting up?
- Where could we introduce agentic AI to make the bottleneck loop faster and more objective?
- As teams become smaller, there will be more opportunities to integrate the Elon Code into workflows.
You don’t have to adopt all of the Elon Code. But even small moves toward hands-on leadership, truth at the source, and deliberate bottleneck hunting can radically improve how a biotech organisation learns and executes.
#Leadership #Management #Innovation #ElonMusk #Founders #Biotech